Coordonnées
patrick.ayotte@usherbrooke.ca
Bureau : D1-2127-2
Laboratoire : D1-1121
Téléphone : (819) 821-7889
Formation
1995 : Maîtrise en Médecine nucléaire et radiobiologie, Université de Sherbrooke
1999 : Doctorat en Chimie, Université de Yale
2001 : Postdoctorat en chimie, Pacific Northwest National Laboratory
Liste des publications (liste complète)
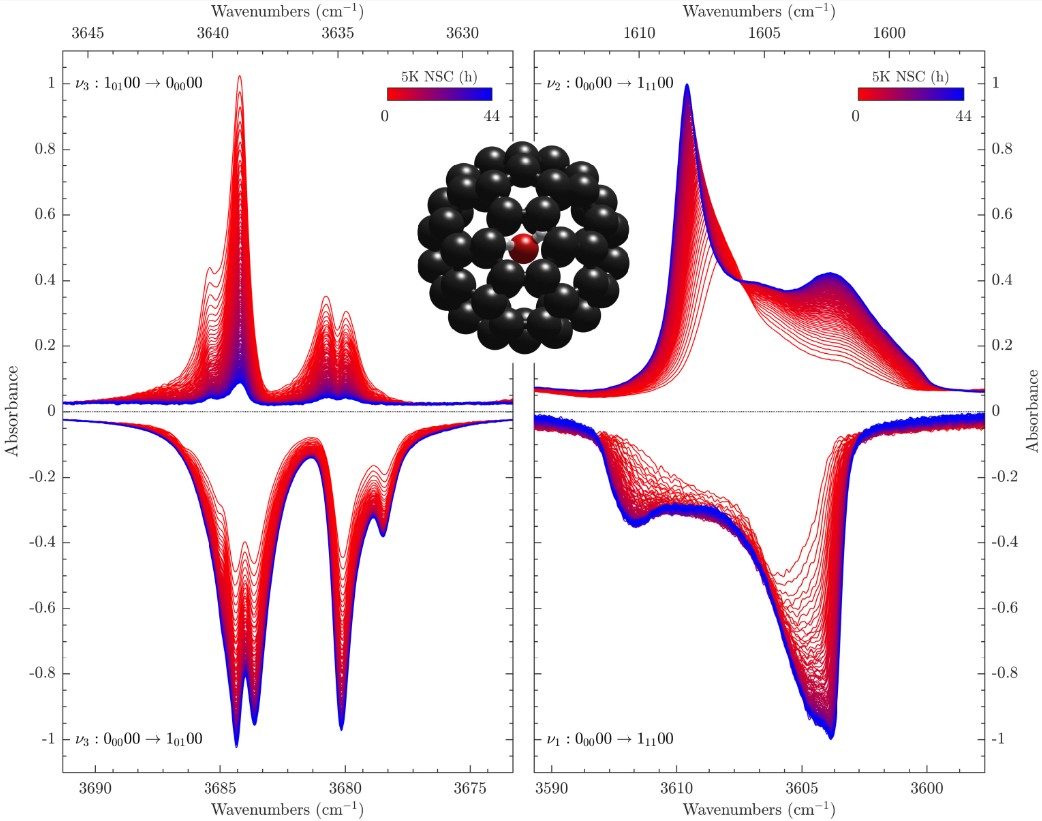
J.-C. Chartrand; T. Putaud; G. Bélanger; M. Bertin; J.-H. Fillion; P. Léveillé; X. Michaut; P. Ayotte
Signatures of rotation–translation couplings, symmetry-breaking, and intermolecular interactions in the rovibrational spectra of solid H2O@C60 Article de journal
Dans: vol. 162, no. 14, 2025, ISSN: 1089-7690.
@article{Chartrand2025,
title = {Signatures of rotation–translation couplings, symmetry-breaking, and intermolecular interactions in the rovibrational spectra of solid H_{2}O@C_{60}},
author = {J.-C. Chartrand and T. Putaud and G. Bélanger and M. Bertin and J.-H. Fillion and P. Léveillé and X. Michaut and P. Ayotte},
doi = {10.1063/5.0253539},
issn = {1089-7690},
year = {2025},
date = {2025-04-14},
urldate = {2025-04-14},
volume = {162},
number = {14},
publisher = {AIP Publishing},
abstract = {Some spectral features observed in the rovibrational spectra of solid H2O@C60 are shown to provide spectroscopic signatures of confinement-induced perturbations related to the coupling between the orientational and positional degrees-of-freedom of the water molecules. Their attribution to either para-H2O@C60 or ortho-H2O@C60 is established from their behavior during nuclear spin conversion. The frequency of the rovibrational transitions that emanate from their ground ro-translational (RT) states appears conspicuously redshifted from that of the corresponding transitions in the free water molecule in the gas phase. However, a few of the 21 hot band spectral features, and one ground state transition, observed in the infrared spectrum of solid H2O@C60 and reported here for the first time, cannot be straightforwardly assigned based on the softening of its intramolecular HOH bending and OH stretching vibrational modes due to confinement within C60. The most strongly perturbed transitions provide insights into the complex confinement-induced quantum nuclear dynamics arising from rotation–translation coupling, allowing the topology of the confinement potential to be revealed using a simple confined rotor model [Putaud et al., J. Chem. Phys. 162, 144313 (2025)]. While the line profiles exhibited by most of the transitions are consistent with symmetry-breaking interactions arising from merohedral disorder in solid H2O@C60, evidence for additional perturbations of the 10100 RT state, in the ground and vibrationally excited manifolds, is reported. Moreover, the line profiles displayed by the transitions emanating from the ground RT state of para-H2O@C60 and the observation of nominally forbidden Q-branch transitions, in the intramolecular HOH bending and symmetric OH stretching ranges of solid H2O@C60 samples with a fill ratio of 75%, are shown to provide a spectroscopic signature of intermolecular dipolar interactions between nearest-neighbor H2O@C60 molecules.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
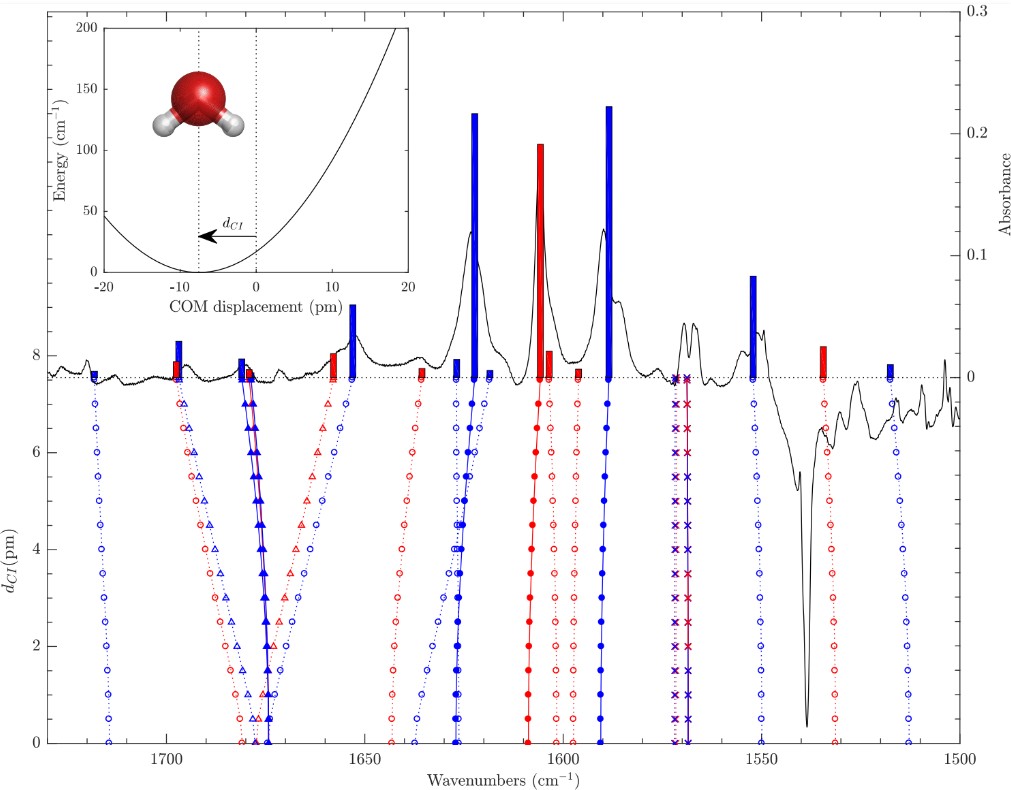
T. Putaud; J.-C. Chartrand; Y. Kalugina; X. Michaut; P.-N. Roy; P. Ayotte
A simple confined rotor model to describe the ro-translational dynamics of water endofullerenes and to assign the ro-vibrational spectra of solid H2O@C60 Article de journal
Dans: vol. 162, no. 14, 2025, ISSN: 1089-7690.
@article{Putaud2025,
title = {A simple confined rotor model to describe the ro-translational dynamics of water endofullerenes and to assign the ro-vibrational spectra of solid H_{2}O@C_{60}},
author = {T. Putaud and J.-C. Chartrand and Y. Kalugina and X. Michaut and P.-N. Roy and P. Ayotte},
doi = {10.1063/5.0253540},
issn = {1089-7690},
year = {2025},
date = {2025-04-14},
urldate = {2025-04-14},
volume = {162},
number = {14},
publisher = {AIP Publishing},
abstract = {A simple Confined Rotor Model (CRM) is used to assign the thirty-nine ro-vibrational transitions observed through the HOH bending and OH stretching ranges of the solid H2O@C60 mid-infrared (MIR) spectra reported in the companion paper [Chartrand et al., J. Chem. Phys. 162, 144312 (2025)]. Assuming that the intramolecular vibrations of the water molecules are separable from their rotational and translational motions, the CRM Hamiltonian describes confinement of H2O within C60 as an eccentric, but otherwise isotropic, 3D harmonic oscillator and as an asymmetric rigid rotor. The topology of the effective confinement potential is constrained using seven transitions observed in the HOH bending range of the MIR spectra of solid H2O@C60, yielding an effective force constant, k = (11.86 ± 0.03) J m−2, and an eccentricity, dCI = (7.55 ± 0.07) pm, in good agreement with DF-LMP2/cc-pVDZ results. While twenty-one broad and overlapping spectral features arising from hot band transitions were described and tentatively assigned by Chartrand et al., some of them appear very strongly perturbed compared to the gas phase. Using the CRM, it is shown that the conspicuous shifts displayed by certain very specific pairs of ro-vibrational transitions provide evidence for confinement-induced rotation–translation coupling between the orientational and positional degrees-of-freedom of endohedral water, resulting in a strong mixing between very specific pairs of ro-translational eigenstates of translational and rotational character. The CRM is shown to provide a satisfactory description of all observed ro-vibrational transitions along with a compelling interpretation for the complex confinement-induced quantum nuclear dynamics of endohedral water as revealed by the rotational and ro-vibrational spectra of solid H2O@C60.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
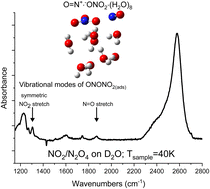
Josée Maurais; Clément Wespiser; Raphaël Robidas; Claude Y. Legault; Patrick Ayotte
Trapping intermediates of the NO2 hydrolysis reaction on ice Article de journal
Dans: Faraday Discuss., vol. 258, p. 546–567, 2025, ISSN: 1364-5498.
@article{Maurais2025,
title = {Trapping intermediates of the NO_{2} hydrolysis reaction on ice},
author = {Josée Maurais and Clément Wespiser and Raphaël Robidas and Claude Y. Legault and Patrick Ayotte},
doi = {10.1039/d4fd00161c},
issn = {1364-5498},
year = {2025},
date = {2025-00-00},
urldate = {2025-00-00},
journal = {Faraday Discuss.},
volume = {258},
pages = {546--567},
publisher = {Royal Society of Chemistry (RSC)},
abstract = {Using molecular beam methods, a mixture of stable NO2, O2NNO2, and up to 30% relative abundance of metastable t-ONONO2, a potential heterogeneous hydrolysis reaction intermediate, was prepared by heating the quasi-effusive molecular beam nozzle to 600 K. The chemical speciation of hot nitrogen dioxide vapours was established using reflection–absorption IR spectroscopy (RAIRS) at very high (i.e., 1 : 1000) dilution by exploiting selective enhancement in absorbance features due to electric field standing waves (EFSW). Mode-selective shifts in the NO stretching vibrational frequencies of these species are observed upon their adsorption to the surface of amorphous solid water (ASW) at 40 K compared to their value in a crystalline solid argon matrix. Their sensitivities to hydration were assessed by computational chemistry methods using clusters of up to ten water molecules. This revealed that the shifts in the vibrational frequency of the terminal N[double bond, length as m-dash]O stretching mode and of the asymmetric ONO stretching mode of the terminal –NO2 group of t-ONONO2 upon its adsorption onto the surface of ASW signal that its ON–ONO2 bond is significantly polarized. Upon thermal annealing of the sample to 130 K, spectral signatures attributed to adsorbed nitrate anions can be observed suggesting that the activation barrier to heterogenous hydrolysis of the O[double bond, length as m-dash]N+·−ONO2 zwitterionic reaction intermediate is sufficiently small to be overcome at cryogenic temperatures. Facile NO2 hydrolysis on aqueous interfaces could contribute to their acidification and to elevated nitrous acid emission fluxes to the lower troposphere.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
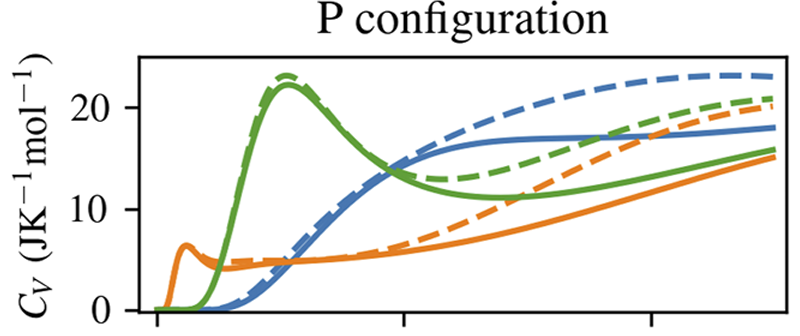
Tobias Serwatka; Spencer Yim; Patrick Ayotte; Pierre-Nicholas Roy
On the nature of the Schottky anomaly in endohedral water Article de journal
Dans: vol. 158, no. 12, 2023, ISSN: 1089-7690.
@article{Serwatka2023,
title = {On the nature of the Schottky anomaly in endohedral water},
author = {Tobias Serwatka and Spencer Yim and Patrick Ayotte and Pierre-Nicholas Roy},
doi = {10.1063/5.0148882},
issn = {1089-7690},
year = {2023},
date = {2023-03-28},
urldate = {2023-03-28},
volume = {158},
number = {12},
publisher = {AIP Publishing},
abstract = {In this work, we study the heat capacity contribution of a rigid water molecule encapsulated in C60 by performing six-dimensional eigenstate calculations with the inclusion of its quantized rotational and translational degrees of freedom. Two confinement model potentials are considered: in the first, confinement is described using distributed pairwise Lennard-Jones interactions, while in the second, the water molecule is trapped within an eccentric but isotropic 3D harmonic effective confinement potential [Wespiser et al., J. Chem. Phys. 156, 074304 (2022)]. Contributions to the heat capacity from both the ortho and para nuclear spin isomers of water are considered to enable the effects of their interconversion to be assessed. By including a symmetry-breaking quadrupolar potential energy term in the Hamiltonian, we can reproduce the experimentally observed Schottky anomaly at ∼2 K [Suzuki et al., J. Phys. Chem. Lett. 10, 1306 (2019)]. Furthermore, our calculations predict a second Schottky anomaly at ∼0.1 K resulting from the H configuration, a different orientational arrangement of the fullerene cages in crystalline solid C60. Contributions from the H configuration to CV also explain the second peak observed at ∼7 K in the experimentally measured heat capacity.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Josée Maurais; Clément Wespiser; Heon Kang; Patrick Ayotte
Preparation and Characterization of Metastable trans-Dinitrogen Tetroxide Article de journal
Dans: J. Phys. Chem. A, vol. 126, no. 15, p. 2353–2360, 2022, ISSN: 1520-5215.
@article{Maurais2022,
title = {Preparation and Characterization of Metastable \textit{trans}-Dinitrogen Tetroxide},
author = {Josée Maurais and Clément Wespiser and Heon Kang and Patrick Ayotte},
doi = {10.1021/acs.jpca.2c01009},
issn = {1520-5215},
year = {2022},
date = {2022-04-21},
urldate = {2022-04-21},
journal = {J. Phys. Chem. A},
volume = {126},
number = {15},
pages = {2353--2360},
publisher = {American Chemical Society (ACS)},
abstract = {Under atmospheric conditions, NO2 is in equilibrium with its dimers, N2O4, which can exist in the form of constitutional isomers and stereoisomers whose relative stabilities and reactivities are still being debated. Experimental limitations facing the spectroscopic characterization of the isomers of N2O4 prevent us from determining their relative contributions to reaction mechanisms possibly causing discrepancies in the reported reaction orders and rates. Using reflection-absorption infrared spectroscopy, molecular beam deposition, and matrix isolation techniques, it is shown that the relative abundances of NO2 and its dimers can be controlled by heating or cooling the deposited gas. The comparison of spectra acquired from samples prepared using molecular beam deposition with those obtained using tube dosing deposition demonstrates how the N2O4 isomer distributions are sensitive to details of the experimental conditions and sample preparation protocols. These observations not only provide a better understanding of a possible source for the disagreements found in the literature, but also a methodology to control and quantify the chemical speciation in NO2 vapors in terms of the relative abundances of NO2 and of the various isomers of N2O4.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}