2025
Putaud, T.; Chartrand, J. -C.; Kalugina, Y.; Michaut, X.; Roy, P. -N.; Ayotte, P.
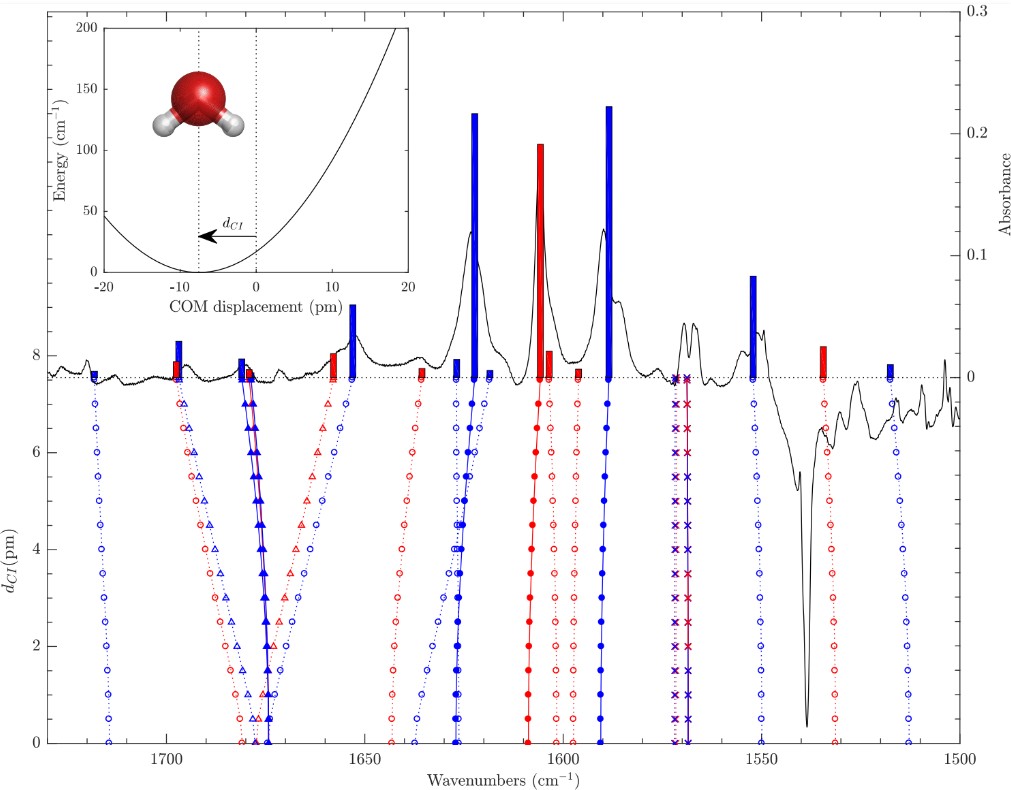
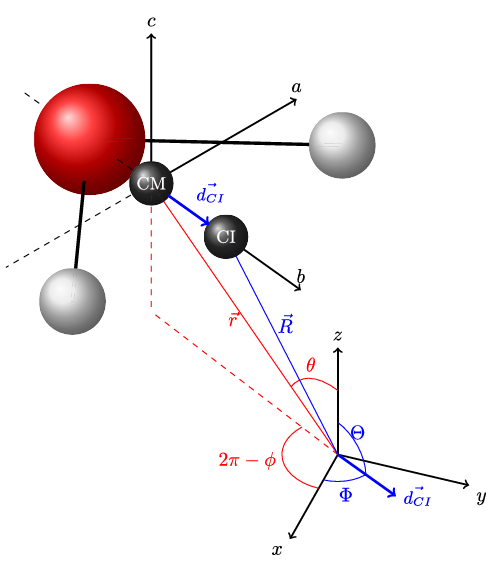
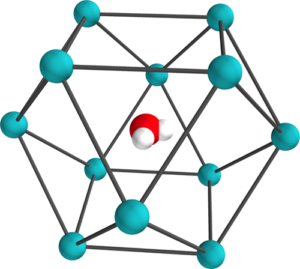
A simple confined rotor model to describe the ro-translational dynamics of water endofullerenes and to assign the ro-vibrational spectra of solid H2O@C60 Article de journal
Dans: vol. 162, no. 14, 2025, ISSN: 1089-7690.
@article{Putaud2025,
title = {A simple confined rotor model to describe the ro-translational dynamics of water endofullerenes and to assign the ro-vibrational spectra of solid H_{2}O@C_{60}},
author = {T. Putaud and J.-C. Chartrand and Y. Kalugina and X. Michaut and P.-N. Roy and P. Ayotte},
doi = {10.1063/5.0253540},
issn = {1089-7690},
year = {2025},
date = {2025-04-14},
urldate = {2025-04-14},
volume = {162},
number = {14},
publisher = {AIP Publishing},
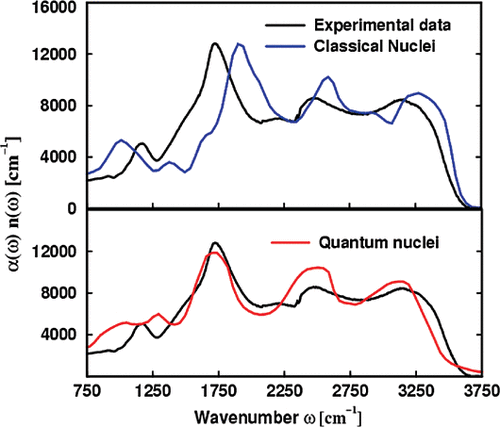
abstract = {A simple Confined Rotor Model (CRM) is used to assign the thirty-nine ro-vibrational transitions observed through the HOH bending and OH stretching ranges of the solid H2O@C60 mid-infrared (MIR) spectra reported in the companion paper [Chartrand et al., J. Chem. Phys. 162, 144312 (2025)]. Assuming that the intramolecular vibrations of the water molecules are separable from their rotational and translational motions, the CRM Hamiltonian describes confinement of H2O within C60 as an eccentric, but otherwise isotropic, 3D harmonic oscillator and as an asymmetric rigid rotor. The topology of the effective confinement potential is constrained using seven transitions observed in the HOH bending range of the MIR spectra of solid H2O@C60, yielding an effective force constant, k = (11.86 ± 0.03) J m−2, and an eccentricity, dCI = (7.55 ± 0.07) pm, in good agreement with DF-LMP2/cc-pVDZ results. While twenty-one broad and overlapping spectral features arising from hot band transitions were described and tentatively assigned by Chartrand et al., some of them appear very strongly perturbed compared to the gas phase. Using the CRM, it is shown that the conspicuous shifts displayed by certain very specific pairs of ro-vibrational transitions provide evidence for confinement-induced rotation–translation coupling between the orientational and positional degrees-of-freedom of endohedral water, resulting in a strong mixing between very specific pairs of ro-translational eigenstates of translational and rotational character. The CRM is shown to provide a satisfactory description of all observed ro-vibrational transitions along with a compelling interpretation for the complex confinement-induced quantum nuclear dynamics of endohedral water as revealed by the rotational and ro-vibrational spectra of solid H2O@C60.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
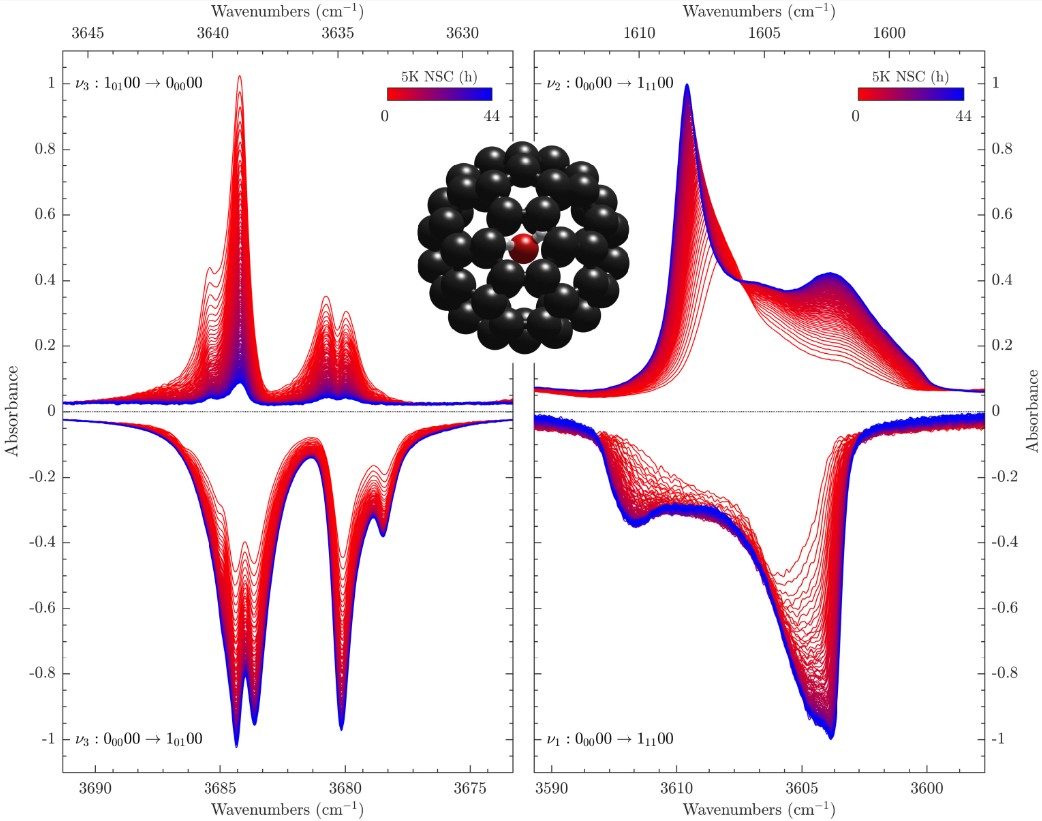
Chartrand, J. -C.; Putaud, T.; Bélanger, G.; Bertin, M.; Fillion, J. -H.; Léveillé, P.; Michaut, X.; Ayotte, P.
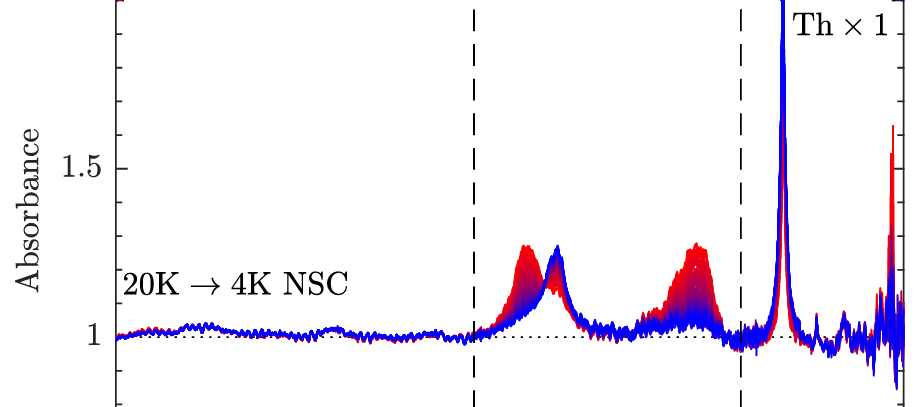
Signatures of rotation–translation couplings, symmetry-breaking, and intermolecular interactions in the rovibrational spectra of solid H2O@C60 Article de journal
Dans: vol. 162, no. 14, 2025, ISSN: 1089-7690.
@article{Chartrand2025,
title = {Signatures of rotation–translation couplings, symmetry-breaking, and intermolecular interactions in the rovibrational spectra of solid H_{2}O@C_{60}},
author = {J.-C. Chartrand and T. Putaud and G. Bélanger and M. Bertin and J.-H. Fillion and P. Léveillé and X. Michaut and P. Ayotte},
doi = {10.1063/5.0253539},
issn = {1089-7690},
year = {2025},
date = {2025-04-14},
urldate = {2025-04-14},
volume = {162},
number = {14},
publisher = {AIP Publishing},
abstract = {Some spectral features observed in the rovibrational spectra of solid H2O@C60 are shown to provide spectroscopic signatures of confinement-induced perturbations related to the coupling between the orientational and positional degrees-of-freedom of the water molecules. Their attribution to either para-H2O@C60 or ortho-H2O@C60 is established from their behavior during nuclear spin conversion. The frequency of the rovibrational transitions that emanate from their ground ro-translational (RT) states appears conspicuously redshifted from that of the corresponding transitions in the free water molecule in the gas phase. However, a few of the 21 hot band spectral features, and one ground state transition, observed in the infrared spectrum of solid H2O@C60 and reported here for the first time, cannot be straightforwardly assigned based on the softening of its intramolecular HOH bending and OH stretching vibrational modes due to confinement within C60. The most strongly perturbed transitions provide insights into the complex confinement-induced quantum nuclear dynamics arising from rotation–translation coupling, allowing the topology of the confinement potential to be revealed using a simple confined rotor model [Putaud et al., J. Chem. Phys. 162, 144313 (2025)]. While the line profiles exhibited by most of the transitions are consistent with symmetry-breaking interactions arising from merohedral disorder in solid H2O@C60, evidence for additional perturbations of the 10100 RT state, in the ground and vibrationally excited manifolds, is reported. Moreover, the line profiles displayed by the transitions emanating from the ground RT state of para-H2O@C60 and the observation of nominally forbidden Q-branch transitions, in the intramolecular HOH bending and symmetric OH stretching ranges of solid H2O@C60 samples with a fill ratio of 75%, are shown to provide a spectroscopic signature of intermolecular dipolar interactions between nearest-neighbor H2O@C60 molecules.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
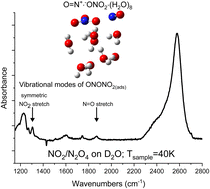
Maurais, Josée; Wespiser, Clément; Robidas, Raphaël; Legault, Claude Y.; Ayotte, Patrick
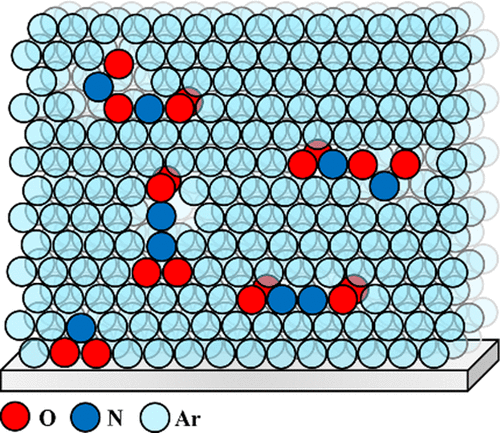
Trapping intermediates of the NO2 hydrolysis reaction on ice Article de journal
Dans: Faraday Discuss., vol. 258, p. 546–567, 2025, ISSN: 1364-5498.
@article{Maurais2025,
title = {Trapping intermediates of the NO_{2} hydrolysis reaction on ice},
author = {Josée Maurais and Clément Wespiser and Raphaël Robidas and Claude Y. Legault and Patrick Ayotte},
doi = {10.1039/d4fd00161c},
issn = {1364-5498},
year = {2025},
date = {2025-00-00},
urldate = {2025-00-00},
journal = {Faraday Discuss.},
volume = {258},
pages = {546--567},
publisher = {Royal Society of Chemistry (RSC)},
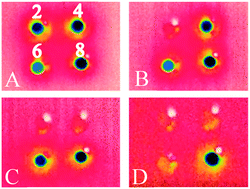
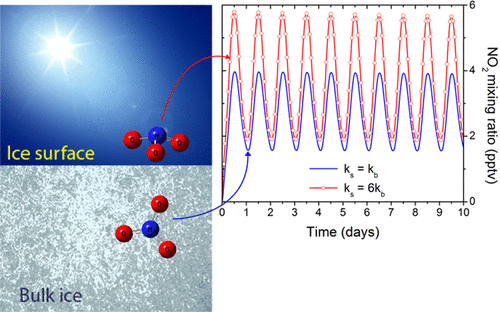
abstract = {Using molecular beam methods, a mixture of stable NO2, O2NNO2, and up to 30% relative abundance of metastable t-ONONO2, a potential heterogeneous hydrolysis reaction intermediate, was prepared by heating the quasi-effusive molecular beam nozzle to 600 K. The chemical speciation of hot nitrogen dioxide vapours was established using reflection–absorption IR spectroscopy (RAIRS) at very high (i.e., 1 : 1000) dilution by exploiting selective enhancement in absorbance features due to electric field standing waves (EFSW). Mode-selective shifts in the NO stretching vibrational frequencies of these species are observed upon their adsorption to the surface of amorphous solid water (ASW) at 40 K compared to their value in a crystalline solid argon matrix. Their sensitivities to hydration were assessed by computational chemistry methods using clusters of up to ten water molecules. This revealed that the shifts in the vibrational frequency of the terminal N[double bond, length as m-dash]O stretching mode and of the asymmetric ONO stretching mode of the terminal –NO2 group of t-ONONO2 upon its adsorption onto the surface of ASW signal that its ON–ONO2 bond is significantly polarized. Upon thermal annealing of the sample to 130 K, spectral signatures attributed to adsorbed nitrate anions can be observed suggesting that the activation barrier to heterogenous hydrolysis of the O[double bond, length as m-dash]N+·−ONO2 zwitterionic reaction intermediate is sufficiently small to be overcome at cryogenic temperatures. Facile NO2 hydrolysis on aqueous interfaces could contribute to their acidification and to elevated nitrous acid emission fluxes to the lower troposphere.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
2023
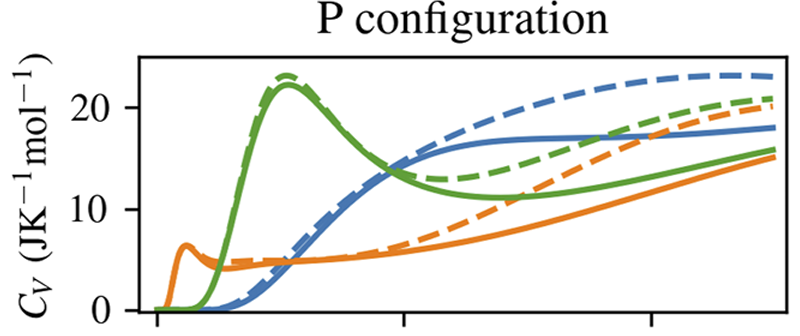
Serwatka, Tobias; Yim, Spencer; Ayotte, Patrick; Roy, Pierre-Nicholas
On the nature of the Schottky anomaly in endohedral water Article de journal
Dans: vol. 158, no. 12, 2023, ISSN: 1089-7690.
@article{Serwatka2023,
title = {On the nature of the Schottky anomaly in endohedral water},
author = {Tobias Serwatka and Spencer Yim and Patrick Ayotte and Pierre-Nicholas Roy},
doi = {10.1063/5.0148882},
issn = {1089-7690},
year = {2023},
date = {2023-03-28},
urldate = {2023-03-28},
volume = {158},
number = {12},
publisher = {AIP Publishing},
abstract = {In this work, we study the heat capacity contribution of a rigid water molecule encapsulated in C60 by performing six-dimensional eigenstate calculations with the inclusion of its quantized rotational and translational degrees of freedom. Two confinement model potentials are considered: in the first, confinement is described using distributed pairwise Lennard-Jones interactions, while in the second, the water molecule is trapped within an eccentric but isotropic 3D harmonic effective confinement potential [Wespiser et al., J. Chem. Phys. 156, 074304 (2022)]. Contributions to the heat capacity from both the ortho and para nuclear spin isomers of water are considered to enable the effects of their interconversion to be assessed. By including a symmetry-breaking quadrupolar potential energy term in the Hamiltonian, we can reproduce the experimentally observed Schottky anomaly at ∼2 K [Suzuki et al., J. Phys. Chem. Lett. 10, 1306 (2019)]. Furthermore, our calculations predict a second Schottky anomaly at ∼0.1 K resulting from the H configuration, a different orientational arrangement of the fullerene cages in crystalline solid C60. Contributions from the H configuration to CV also explain the second peak observed at ∼7 K in the experimentally measured heat capacity.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
2022
Maurais, Josée; Wespiser, Clément; Kang, Heon; Ayotte, Patrick
Preparation and Characterization of Metastable trans-Dinitrogen Tetroxide Article de journal
Dans: J. Phys. Chem. A, vol. 126, no. 15, p. 2353–2360, 2022, ISSN: 1520-5215.
@article{Maurais2022,
title = {Preparation and Characterization of Metastable \textit{trans}-Dinitrogen Tetroxide},
author = {Josée Maurais and Clément Wespiser and Heon Kang and Patrick Ayotte},
doi = {10.1021/acs.jpca.2c01009},
issn = {1520-5215},
year = {2022},
date = {2022-04-21},
urldate = {2022-04-21},
journal = {J. Phys. Chem. A},
volume = {126},
number = {15},
pages = {2353--2360},
publisher = {American Chemical Society (ACS)},
abstract = {Under atmospheric conditions, NO2 is in equilibrium with its dimers, N2O4, which can exist in the form of constitutional isomers and stereoisomers whose relative stabilities and reactivities are still being debated. Experimental limitations facing the spectroscopic characterization of the isomers of N2O4 prevent us from determining their relative contributions to reaction mechanisms possibly causing discrepancies in the reported reaction orders and rates. Using reflection-absorption infrared spectroscopy, molecular beam deposition, and matrix isolation techniques, it is shown that the relative abundances of NO2 and its dimers can be controlled by heating or cooling the deposited gas. The comparison of spectra acquired from samples prepared using molecular beam deposition with those obtained using tube dosing deposition demonstrates how the N2O4 isomer distributions are sensitive to details of the experimental conditions and sample preparation protocols. These observations not only provide a better understanding of a possible source for the disagreements found in the literature, but also a methodology to control and quantify the chemical speciation in NO2 vapors in terms of the relative abundances of NO2 and of the various isomers of N2O4.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Wespiser, Clément; Putaud, Thomas; Kalugina, Yulia; Soldera, Armand; Roy, Pierre-Nicholas; Michaut, Xavier; Ayotte, Patrick
Ro-translational dynamics of confined water. I. The confined asymmetric rotor model Article de journal
Dans: vol. 156, no. 7, 2022, ISSN: 1089-7690.
@article{Wespiser2022,
title = {Ro-translational dynamics of confined water. I. The confined asymmetric rotor model},
author = {Clément Wespiser and Thomas Putaud and Yulia Kalugina and Armand Soldera and Pierre-Nicholas Roy and Xavier Michaut and Patrick Ayotte},
doi = {10.1063/5.0079565},
issn = {1089-7690},
year = {2022},
date = {2022-02-21},
urldate = {2022-02-21},
volume = {156},
number = {7},
publisher = {AIP Publishing},
abstract = {Confinement effects on the ro-translational (RT) dynamics of water, trapped in rare gas matrices or within endofullerenes (i.e., H2O@C60), can be experimentally assessed using rotationally resolved far-infrared, or mid-infrared, spectroscopy [Putaud et al., J. Chem. Phys. 156, 074305 (2022) (Paper II)]. The confined rotor model is used here to reveal how the quantized rotational and frustrated translational energy levels of confined water interact and mix by way of the confinement-induced rotation-translation coupling (RTC). An eccentric but otherwise isotropic 3D harmonic effective potential is used to account for confinement effects, thereby allowing the dependence of the magnitude of the RTC on the topology of the model confinement potential, the resulting intricate mixing schemes, and their impact on the RT energy levels to be examined in detail. The confined rotor model thus provides a convenient framework to investigate the matrix and isotope effects on the RT dynamics of water under extreme confinement probed spectroscopically, thereby potentially providing insight into the mechanisms and rates for ortho-H2O ↔ para-H2O nuclear spin isomer interconversion in confined water.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Putaud, T.; Wespiser, C.; Bertin, M.; Fillion, J. -H.; Kalugina, Y.; Jeseck, P.; Milpanis, A.; Philippe, L.; Soulard, P.; Tremblay, B.; Tuloup, C.; Ayotte, P.; Michaut, X.
Dans: vol. 156, no. 7, 2022, ISSN: 1089-7690.
@article{Putaud2022,
title = {Rotranslational dynamics of confined water. II. Spectroscopic evidence of confinement effects on the far-infrared spectra of water isotopologues in argon and krypton matrices},
author = {T. Putaud and C. Wespiser and M. Bertin and J.-H. Fillion and Y. Kalugina and P. Jeseck and A. Milpanis and L. Philippe and P. Soulard and B. Tremblay and C. Tuloup and P. Ayotte and X. Michaut},
doi = {10.1063/5.0079566},
issn = {1089-7690},
year = {2022},
date = {2022-02-21},
urldate = {2022-02-21},
volume = {156},
number = {7},
publisher = {AIP Publishing},
abstract = {Water molecules trapped in rare gas matrices exhibit conspicuous shifts in their far-infrared (FIR), rotranslational spectral features compared with the corresponding transitions observed in the gas phase. These confinement-induced perturbations have been related not only to the quantization of translational motion but also to the coupling between the orientational and positional degrees of freedom: the rotation–translation coupling (RTC). As the propensity displayed by the nuclear spin isomers (NSI) of water to undergo interconversion in confinement is intimately related to how its nuclear spin degrees of freedom are coupled with those for intra- and intermolecular motions, confinement-induced RTC should also strongly impact the NSI interconversion mechanisms and rates. Insight into the rotranslational dynamics for H216O, H217O, and H218O, confined in argon and krypton matrices, is provided here based on the evolution of rotranslational spectra induced by NSI interconversion while a definitive assignment is provided from the transition energies and intensities calculated using the confined rotor model [Paper I, Wespiser et al., J. Chem. Phys. 156, 074304 (2021)]. In order to build a complete rotranslational energy diagram of confined water, which is fundamental to understand the NSI interconversion rates, the energy difference between the ground ortho and para rotranslational states is derived from the temperature dependence of the intensity ratio of mid-infrared lines emerging from these states. These investigations should provide deeper insight of the factors that control NSI interconversion of water isotopologues under extreme confinement.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
2021
Wespiser, Clément; Ayotte, Patrick; Soldera, Armand
Dans: Molecular Simulation, vol. 47, no. 10-11, p. 942–949, 2021, ISSN: 1029-0435.
@article{Wespiser2020,
title = {Exploring rotation-translation coupling for a confined asymmetric rotor using molecular dynamics simulations: the case of the water molecule trapped inside a rare gas matrix},
author = {Clément Wespiser and Patrick Ayotte and Armand Soldera},
doi = {10.1080/08927022.2020.1807019},
issn = {1029-0435},
year = {2021},
date = {2021-07-24},
urldate = {2021-07-24},
journal = {Molecular Simulation},
volume = {47},
number = {10-11},
pages = {942--949},
publisher = {Informa UK Limited},
abstract = {Differences between gas-phase and matrix-isolated rotational and rovibrational spectra of the water molecule are interpreted in term of the confined rotor model. The parameters of this model enable the isotopic composition of the molecule, which has non-trivial impacts on the spectra of matrix-isolated confined rotors, to be taken into account on a very simple and intuitive basis. We use molecular dynamics simulations to systematically explore the effects of the mass distribution of various isotopomers of the water molecule on the coupled rotational and translational dynamics of the confined asymmetric rotor, and on their coupling with the phonons of the argon matrix. Analysis of the trajectories reveals that, depending on the mass distribution, a preferred orientation of the water molecule can be strongly imposed by the topology of its interaction potential with the confinement medium. Features of the confining potential, and of the rotation-translation coupling, are thus revealed from classical molecular dynamics simulations.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Maurais, Josée; Orban, Frédéric; Dauphinais, Emrik; Ayotte, Patrick
Monitoring moisture content and evaporation kinetics from mine slurries through albedo measurements to help predict and prevent dust emissions Article de journal
Dans: R. Soc. open sci., vol. 8, no. 7, 2021, ISSN: 2054-5703.
@article{Maurais2021,
title = {Monitoring moisture content and evaporation kinetics from mine slurries through albedo measurements to help predict and prevent dust emissions},
author = {Josée Maurais and Frédéric Orban and Emrik Dauphinais and Patrick Ayotte},
doi = {10.1098/rsos.210414},
issn = {2054-5703},
year = {2021},
date = {2021-07-00},
urldate = {2021-07-00},
journal = {R. Soc. open sci.},
volume = {8},
number = {7},
publisher = {The Royal Society},
abstract = {The prediction and prevention of fugitive dust emissions from mine tailings surfaces depend largely on our ability to monitor and monitor and predict the evolution of tailings moisture content (TMC). Albedo measurements are demonstrated here to be valuable tools to quantify TMC in bauxite residue samples under controlled conditions in the laboratory. The difference in albedo between 1.30 and 1.55 µm obtained through the infrared integrating sphere method shows good correlations with those acquired with a field spectroradiometer while both are strongly correlated with TMC. Additionally, continuous spectroscopic characterization of evaporating residues is shown to reveal the evolution in their surface drying rates. These optical methods could help predict surface drying state, thereby improving the accuracy of dust emissions risk assessment protocols that support mining industries intervention and mitigation strategies.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
2020
Maurais, Josée; Ayotte, Patrick
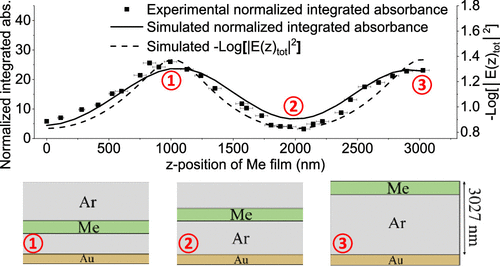
Tailoring Electric Field Standing Waves in Reflection–Absorption Infrared Spectroscopy to Enhance Absorbance in Buried Molecular Layers Article de journal
Dans: J. Phys. Chem. C, vol. 124, no. 31, p. 17158–17164, 2020, ISSN: 1932-7455.
@article{Maurais2020,
title = {Tailoring Electric Field Standing Waves in Reflection–Absorption Infrared Spectroscopy to Enhance Absorbance in Buried Molecular Layers},
author = {Josée Maurais and Patrick Ayotte},
doi = {10.1021/acs.jpcc.0c05309},
issn = {1932-7455},
year = {2020},
date = {2020-08-06},
urldate = {2020-08-06},
journal = {J. Phys. Chem. C},
volume = {124},
number = {31},
pages = {17158--17164},
publisher = {American Chemical Society (ACS)},
abstract = {Electric field standing waves (EFSW) that establish within thin films during their characterization by reflection–absorption infrared (RAIR) spectroscopy are shown to result in a >25-fold enhancement in the intensity of absorbance features in a molecularly thin methane (Me) layer buried within Argon (Ar) films. Using an all-optical depth profiling method and describing the interference effects responsible for these phenomena using a classical optics model, modulations in the EFSW amplitude at specific sample thicknesses are demonstrated to be responsible for these strong deviations from Beer–Lambert behavior. Tuning sample thickness is shown to enable the EFSW depth profile to be tailored, conferring greatly enhanced sensitivity and increased spatial selectivity to RAIR spectroscopy. These are highly desirable attributes for the characterization of interfacial structures and mesoscopic dynamic processes within complex buried multilayer assemblies and stratified composite films used to model and study a range of materials and life science phenomena.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Maurais, Josée; Beaumont, Étienne; Bourret, Joanick; Dauphinais, Emrik; Bouchard, Nicolas-Alexandre; Ayotte, Patrick
A thermal imaging methodology to study evaporation kinetics in mine tailings Article de journal
Dans: Environ. Sci.: Water Res. Technol., vol. 6, no. 5, p. 1456–1464, 2020, ISSN: 2053-1419.
@article{Maurais2020c,
title = {A thermal imaging methodology to study evaporation kinetics in mine tailings},
author = {Josée Maurais and Étienne Beaumont and Joanick Bourret and Emrik Dauphinais and Nicolas-Alexandre Bouchard and Patrick Ayotte},
doi = {10.1039/d0ew00104j},
issn = {2053-1419},
year = {2020},
date = {2020-05-07},
urldate = {2020-05-07},
journal = {Environ. Sci.: Water Res. Technol.},
volume = {6},
number = {5},
pages = {1456--1464},
publisher = {Royal Society of Chemistry (RSC)},
abstract = {Predicting why, how, and when mine tailings disposal sites become prone to dust scattering events is often hampered by our limited understanding of the factors that affect the drying rates from their surface layers. As a case study, thermal imaging is demonstrated here to be a valuable tool to study the evaporation mechanisms and rates from bauxite residues as a function of their thickness and physicochemical properties, as well as environmental conditions. These investigations reveal that their late stage drying rates are limited by gas phase diffusion through the interstitial air within their internal microporosity. The smallness of the effective diffusion coefficient indicates that water adsorption on bauxite residues surfaces is the dominant phenomenon responsible for their slow water vapour transport kinetics, a phenomenon that ultimately controls their late stage drying rates, that is when dust scattering is most likely to occur. As such, application of this thermal imaging methodology in the field may also contribute to improve the accuracy of risk assessment protocols, support intervention and mitigation strategies, underpin optimization efforts for mining residues management, and improve forecasting of fugitive dust emissions from mine tailings by enabling more accurate predictions of the evolution in their surface drying state.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Maurais, Josée; Ayotte, Patrick
Tailoring electric field standing waves in reflection–absorption infrared spectroscopy to enhance absorbance from adsorbates on ice surfaces Article de journal
Dans: vol. 152, no. 7, 2020, ISSN: 1089-7690.
@article{Maurais2020b,
title = {Tailoring electric field standing waves in reflection–absorption infrared spectroscopy to enhance absorbance from adsorbates on ice surfaces},
author = {Josée Maurais and Patrick Ayotte},
doi = {10.1063/1.5141934},
issn = {1089-7690},
year = {2020},
date = {2020-02-21},
urldate = {2020-02-21},
volume = {152},
number = {7},
publisher = {AIP Publishing},
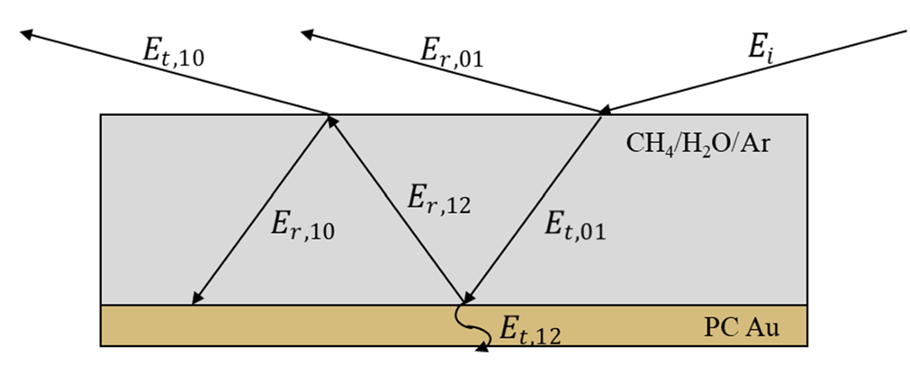
abstract = {The spectroscopic detection of molecules adsorbed onto ice surfaces at coverages similar to those encountered under typical environmental conditions requires high surface selectivity and sensitivity that few techniques can afford. An experimental methodology allowing a significant enhancement in the absorbance from adsorbed molecules is demonstrated herein. It exploits Electric Field Standing Wave (EFSW) effects intrinsic to grazing incidence Reflection–Absorption Infrared (RAIR) spectroscopy, where film thickness dependent optical interferences occur between the multiple reflections of the IR beam at the film–vacuum and the substrate–film interfaces. In this case study, CH4 is used as a probe molecule and is deposited on a 20 ML coverage dense amorphous solid water film adsorbed onto solid Ar underlayers of various thicknesses. We observe that, at thicknesses where destructive interferences coincide with the absorption features from the CH stretching and HCH bending vibrational modes of methane, their intensity increases by a factor ranging from 10 to 25. Simulations of the RAIR spectra of the composite stratified films using a classical optics model reproduce the Ar underlayer coverage dependent enhancements of the absorbance features from CH4 adsorbed onto the ice surface. They also reveal that the enhancements occur when the square modulus of the total electric field at the film’s surface reaches its minimum value. Exploiting the EFSW effect allows the limit of detection to be reduced to a coverage of (0.2 ± 0.2) ML CH4, which opens up interesting perspectives for spectroscopic studies of heterogeneous atmospheric chemistry at coverages that are more representative of those found in the natural environment.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Nagashima, Ken; Maurais, Josée; Murata, Ken-ichiro; Furukawa, Yoshinori; Ayotte, Patrick; Sazaki, Gen
Appearance and Disappearance of Quasi-Liquid Layers on Ice Crystals in the Presence of Nitric Acid Gas Article de journal
Dans: Crystals, vol. 10, no. 2, 2020, ISSN: 2073-4352.
@article{Nagashima2020,
title = {Appearance and Disappearance of Quasi-Liquid Layers on Ice Crystals in the Presence of Nitric Acid Gas},
author = {Ken Nagashima and Josée Maurais and Ken-ichiro Murata and Yoshinori Furukawa and Patrick Ayotte and Gen Sazaki},
doi = {10.3390/cryst10020072},
issn = {2073-4352},
year = {2020},
date = {2020-02-00},
urldate = {2020-02-00},
journal = {Crystals},
volume = {10},
number = {2},
publisher = {MDPI AG},
abstract = {The surfaces of ice crystals near the melting point are covered with thin liquid water layers, called quasi-liquid layers (QLLs), which play crucial roles in various chemical reactions in nature. So far, there have been many spectroscopic studies of such chemical reactions on ice surfaces, however, revealing the effects of atmospheric gases on ice surfaces remains an experimental challenge. In this study, we chose HNO3 as a model atmospheric gas, and directly observed the ice basal faces by advanced optical microscopy under partial pressure of HNO3 (~10−4 Pa), relevant to those found in the atmosphere. We found that droplets (HNO3-QLLs) appeared on ice surfaces at temperatures ranging from −0.9 to −0.2 °C with an increase in temperature, and that they disappeared at temperatures ranging from −0.6 to −1.3 °C with decreasing temperature. We also found that the size of the HNO3-QLLs decreased immediately after we started reducing the temperature. From the changes in size and the liquid–solid phase diagram of the HNO3-H2O binary system, we concluded that the HNO3-QLLs did not consist of pure water, but rather aqueous HNO3 solutions, and that the temperature and HNO3 concentration of the HNO3-QLLs also coincided with those along a liquidus line.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
2019
Kang, Hani; Maurais, Josée; Park, Youngwook; Ayotte, Patrick; Kang, Heon
Electric Field Effect on Condensed-Phase Molecular Systems. VIII. Vibrational Stark Effect and Dipolar Inversion in a Carbon Monoxide Crystal Article de journal
Dans: J. Phys. Chem. C, vol. 123, no. 51, p. 31262–31271, 2019, ISSN: 1932-7455.
@article{Kang2019,
title = {Electric Field Effect on Condensed-Phase Molecular Systems. VIII. Vibrational Stark Effect and Dipolar Inversion in a Carbon Monoxide Crystal},
author = {Hani Kang and Josée Maurais and Youngwook Park and Patrick Ayotte and Heon Kang},
doi = {10.1021/acs.jpcc.9b08902},
issn = {1932-7455},
year = {2019},
date = {2019-12-26},
urldate = {2019-12-26},
journal = {J. Phys. Chem. C},
volume = {123},
number = {51},
pages = {31262--31271},
publisher = {American Chemical Society (ACS)},
abstract = {We applied a strong (≤2.6 × 108 V·m–1) external electric field across a carbon monoxide crystal film at 10 K and studied its effect on the sample with reflection–absorption infrared spectroscopy (RAIRS). The vibrational Stark effect (VSE) on the intramolecular CO stretching vibrations of the minor isotopologues (13C16O and 12C18O) reveal the spectral signature of isolated CO vibrations, decoupled from crystal phonons in the solid, as a function of the external electric field magnitude. These so-called molecular CO bands display a VSE with a sensitivity factor of 0.69 ± 0.05 cm–1/(108 V·m–1) in crystalline CO. The VSE on the coupled CO stretching vibrations of the major isotopologue (12C16O) was measured for crystalline and amorphous solid CO films, and the results were analyzed with the help of a classical optics model of RAIRS for thin solid films. In addition to these spectral changes due to VSE, the external electric field facilitates the head-to-tail inversion of CO dipoles in the crystal lattice as a result of electrostatic interactions. This result is the first experimental demonstration of dipole inversion in a molecular crystal induced by a DC electric field. The dipole inversion occurs slowly and irreversibly in crystalline CO, reaching a yield of up to about 20% dipole inversion at an external field strength of 2.6 × 108 V·m–1 at 10 K. The observed yield of dipole inversion is interpreted in terms of a thermodynamic model that accounts for the electrostatic stabilization energy of dipoles and the configurational entropy of the CO crystal. The present study demonstrates that a polarized CO crystal with reduced residual entropy can be formed by applying a strong electric field at low temperature.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Vermette, Jonathan; Braud, Isabelle; Turgeon, Pierre-Alexandre; Alexandrowicz, Gil; Ayotte, Patrick
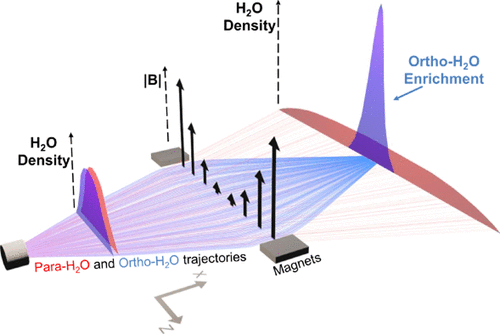
Quantum State-Resolved Characterization of a Magnetically Focused Beam of ortho-H2O Article de journal
Dans: J. Phys. Chem. A, vol. 123, no. 42, p. 9234–9239, 2019, ISSN: 1520-5215.
@article{Vermette2019,
title = {Quantum State-Resolved Characterization of a Magnetically Focused Beam of \textit{ortho}-H_{2}O},
author = {Jonathan Vermette and Isabelle Braud and Pierre-Alexandre Turgeon and Gil Alexandrowicz and Patrick Ayotte},
doi = {10.1021/acs.jpca.9b04294},
issn = {1520-5215},
year = {2019},
date = {2019-10-24},
urldate = {2019-10-24},
journal = {J. Phys. Chem. A},
volume = {123},
number = {42},
pages = {9234--9239},
publisher = {American Chemical Society (ACS)},
abstract = {Magnetic focusing of a molecular beam formed from a rotationally cooled supersonic jet of H2O seeded in argon is shown to yield water vapor highly enriched in the ortho-H2O nuclear spin isomer (NSI). Rotationally resolved resonance-enhanced multiphoton ionization time-of-flight mass spectrometry demonstrates that this methodology enables the preparation of a beam of water molecules enriched to >98% in the ortho-H2O NSI, that is, having an ortho-to-para ratio in excess of 50:1. The flux and quantum-state purity achieved through the methodology described herein could enable heterogeneous chemistry applications including the preparation of nuclear spin-polarized water adlayers, making nuclear magnetic resonance investigations amenable to surface science studies, as well as laboratory astrophysics investigations of NSI interconversion mechanisms and rates in ice and at its surface.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
2018
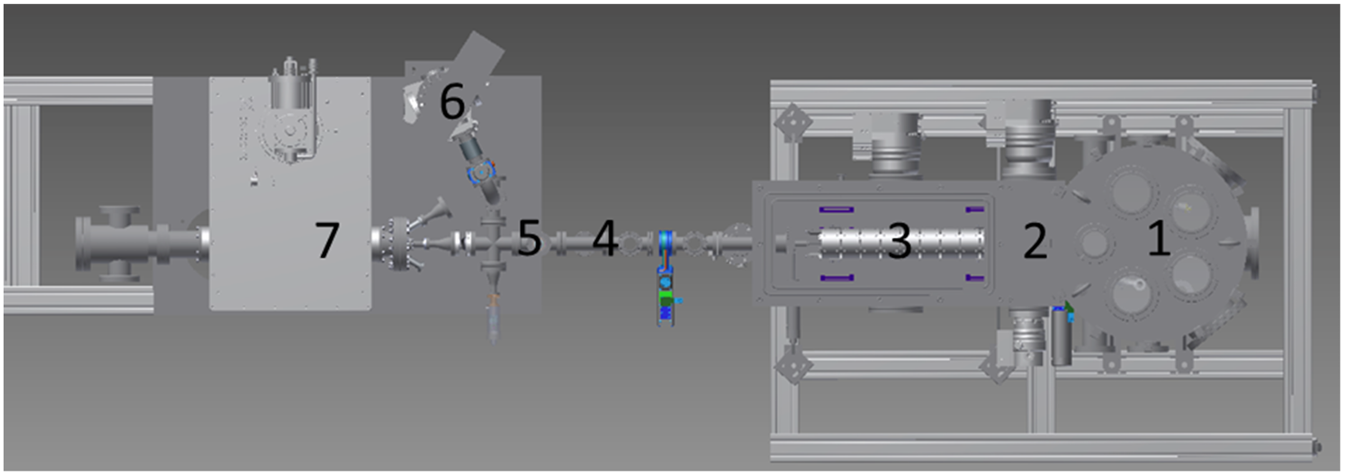
Krüger, C.; Lisitsin-Baranovsky, E.; Ofer, O.; Turgeon, P. -A.; Vermette, J.; Ayotte, P.; Alexandrowicz, G.
A magnetically focused molecular beam source for deposition of spin-polarised molecular surface layers Article de journal
Dans: vol. 149, no. 16, 2018, ISSN: 1089-7690.
@article{Krüger2018,
title = {A magnetically focused molecular beam source for deposition of spin-polarised molecular surface layers},
author = {C. Krüger and E. Lisitsin-Baranovsky and O. Ofer and P.-A. Turgeon and J. Vermette and P. Ayotte and G. Alexandrowicz},
doi = {10.1063/1.5048521},
issn = {1089-7690},
year = {2018},
date = {2018-10-28},
urldate = {2018-10-28},
volume = {149},
number = {16},
publisher = {AIP Publishing},
abstract = {<Separating molecular spin isomers is a challenging task, with potential applications in various fields ranging from astrochemistry to magnetic resonance imaging. A new promising method for spin-isomer separation is magnetic focusing, a method which was shown to be capable of producing a molecular beam of ortho-water. Here, we present results from a modified magnetic focusing apparatus and show that it can be used to separate the spin isomers of acetylene and methane. From the measured focused profiles of the molecular beams and a numerical simulation analysis, we provide estimations for the spin purity and the significantly improved molecular flux obtained with the new setup. Finally, we discuss the spin-relaxation conditions which will be needed to apply this new source for measuring nuclear magnetic resonance signals of a single surface layer.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
2017
Turgeon, Pierre-Alexandre; Vermette, Jonathan; Alexandrowicz, Gil; Peperstraete, Yoann; Philippe, Laurent; Bertin, Mathieu; Fillion, Jean-Hugues; Michaut, Xavier; Ayotte, Patrick
Confinement Effects on the Nuclear Spin Isomer Conversion of H2O Article de journal
Dans: J. Phys. Chem. A, vol. 121, no. 8, p. 1571–1576, 2017, ISSN: 1520-5215.
@article{Turgeon2017,
title = {Confinement Effects on the Nuclear Spin Isomer Conversion of H_{2}O},
author = {Pierre-Alexandre Turgeon and Jonathan Vermette and Gil Alexandrowicz and Yoann Peperstraete and Laurent Philippe and Mathieu Bertin and Jean-Hugues Fillion and Xavier Michaut and Patrick Ayotte},
doi = {10.1021/acs.jpca.7b00893},
issn = {1520-5215},
year = {2017},
date = {2017-03-02},
urldate = {2017-03-02},
journal = {J. Phys. Chem. A},
volume = {121},
number = {8},
pages = {1571--1576},
publisher = {American Chemical Society (ACS)},
abstract = {The mechanism for interconversion between the nuclear spin isomers (NSI) of H2O remains shrouded in uncertainties. The temperature dependence displayed by NSI interconversion rates for H2O isolated in an argon matrix provides evidence that confinement effects are responsible for the dramatic increase in their kinetics with respect to the gas phase, providing new pathways for o-H2O↔p-H2O conversion in endohedral compounds. This reveals intramolecular aspects of the interconversion mechanism which may improve methodologies for the separation and storage of NSI en route to applications ranging from magnetic resonance spectroscopy and imaging to interpretations of spin temperatures in the interstellar medium.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
2016
Lisitsin-Baranovsky, E.; Delage, S.; Sucre, O.; Ofer, O.; Ayotte, P.; Alexandrowicz, G.
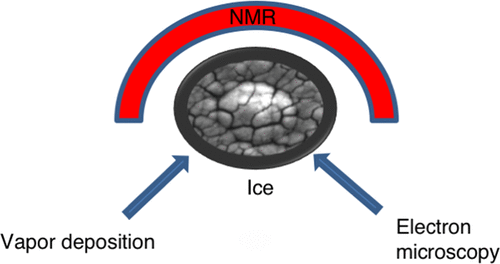
In Situ NMR Measurements of Vapor Deposited Ice Article de journal
Dans: J. Phys. Chem. C, vol. 120, no. 44, p. 25445–25450, 2016, ISSN: 1932-7455.
@article{Lisitsin-Baranovsky2016,
title = {In Situ NMR Measurements of Vapor Deposited Ice},
author = {E. Lisitsin-Baranovsky and S. Delage and O. Sucre and O. Ofer and P. Ayotte and G. Alexandrowicz},
doi = {10.1021/acs.jpcc.6b08746},
issn = {1932-7455},
year = {2016},
date = {2016-11-10},
urldate = {2016-11-10},
journal = {J. Phys. Chem. C},
volume = {120},
number = {44},
pages = {25445--25450},
publisher = {American Chemical Society (ACS)},
abstract = {In situ NMR spin–lattice relaxation measurements were performed on several vapor deposited ices. The measurements, which span more than 6 orders of magnitude in relaxation times, show a complex spin–lattice relaxation pattern that is strongly dependent on the growth conditions of the sample. The relaxation patterns change from multitime scale relaxation for samples grown at temperatures below the amorphous–crystalline transition temperature to single exponential recovery for samples grown above the transition temperature. The slow-relaxation contribution seen in cold-grown samples exhibits a temperature dependence, and becomes even slower after the sample is annealed at 200 K. The fast-relaxation contribution seen in these samples, does not seem to change or disappear even when heating to temperatures where the sample is evaporated. The possibility that the fast relaxation component is linked to the microporous structures in amorphous ice samples is further examined using an environmental electron scanning microscope. The images reveal complex mesoscale microporous structures which maintain their morphology up to their desorption temperatures. These findings, support the possibility that water molecules at pore surfaces might be responsible for the fast-relaxation contribution. Furthermore, the results of this study indicate that the pore-collapse dynamics observed in the past in amorphous ices using other experimental techniques, might be effectively inhibited in samples which are grown by relatively fast vapor deposition.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
2015
Marcotte, Guillaume; Marchand, Patrick; Pronovost, Stéphanie; Ayotte, Patrick; Laffon, Carine; Parent, Philippe
Surface-Enhanced Nitrate Photolysis on Ice Article de journal
Dans: J. Phys. Chem. A, vol. 119, no. 10, p. 1996–2005, 2015, ISSN: 1520-5215.
@article{Marcotte2015,
title = {Surface-Enhanced Nitrate Photolysis on Ice},
author = {Guillaume Marcotte and Patrick Marchand and Stéphanie Pronovost and Patrick Ayotte and Carine Laffon and Philippe Parent},
doi = {10.1021/jp511173w},
issn = {1520-5215},
year = {2015},
date = {2015-03-12},
urldate = {2015-03-12},
journal = {J. Phys. Chem. A},
volume = {119},
number = {10},
pages = {1996--2005},
publisher = {American Chemical Society (ACS)},
abstract = {Heterogeneous nitrate photolysis is the trigger for many chemical processes occurring in the polar boundary layer and is widely believed to occur in a quasi-liquid layer (QLL) at the surface of ice. The dipole-forbidden character of the electronic transition relevant to boundary layer atmospheric chemistry and the small photolysis/photoproduct yields in ice (and in water) may confer a significant enhancement and interfacial specificity to this important photochemical reaction at the surface of ice. Using amorphous solid water films at cryogenic temperatures as models for the disordered interstitial air–ice interface within the snowpack suppresses the diffusive uptake kinetics, thereby prolonging the residence time of nitrate anions at the surface of ice. This approach allows their slow heterogeneous photolysis kinetics to be studied, providing the first direct evidence that nitrates adsorbed onto the first molecular layer at the surface of ice are photolyzed more effectively than those dissolved within the bulk. Vibrational spectroscopy allows the ∼3-fold enhancement in photolysis rates to be correlated with the nitrates’ distorted intramolecular geometry, thereby hinting at the role played by the greater chemical heterogeneity in their solvation environment at the surface of ice than that in the bulk. A simple 1D kinetic model suggests (1) that a 3(6)-fold enhancement in photolysis rate for nitrates adsorbed onto the ice surface could increase the photochemical NO2 emissions from a 5(8) nm thick photochemically active interfacial layer by 30(60)%, and (2) that 25(40)% of the NO2 photochemical emissions to the snowpack interstitial air are released from the topmost molecularly thin surface layer on ice. These findings may provide a new paradigm for heterogeneous (photo)chemistry at temperatures below those required for a QLL to form at the ice surface.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
2013
Marcotte, Guillaume; Ayotte, Patrick; Bendounan, Azzedine; Sirotti, Fausto; Laffon, Carine; Parent, Philippe
Dissociative Adsorption of Nitric Acid at the Surface of Amorphous Solid Water Revealed by X-ray Absorption Spectroscopy Article de journal
Dans: J. Phys. Chem. Lett., vol. 4, no. 16, p. 2643–2648, 2013, ISSN: 1948-7185.
@article{Marcotte2013,
title = {Dissociative Adsorption of Nitric Acid at the Surface of Amorphous Solid Water Revealed by X-ray Absorption Spectroscopy},
author = {Guillaume Marcotte and Patrick Ayotte and Azzedine Bendounan and Fausto Sirotti and Carine Laffon and Philippe Parent},
doi = {10.1021/jz401310j},
issn = {1948-7185},
year = {2013},
date = {2013-08-15},
urldate = {2013-08-15},
journal = {J. Phys. Chem. Lett.},
volume = {4},
number = {16},
pages = {2643--2648},
publisher = {American Chemical Society (ACS)},
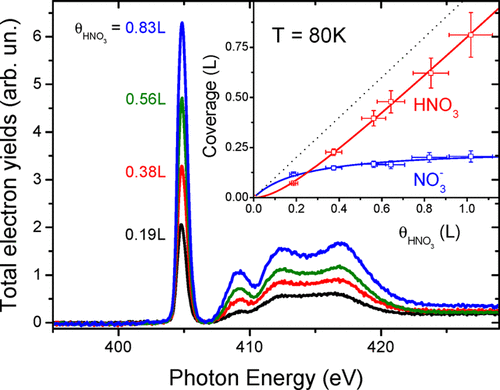
abstract = {A spectral feature unique to the molecularly adsorbed state of HNO3 is found in X-ray absorption spectroscopy. This distinctive signature reveals the extent to which nitric acid is ionically dissociated upon its adsorption on amorphous solid water (ASW) at low coverage and low temperature. Thermal annealing induces irreversible proton transfer from HNO3(ads), demonstrating that it is metastable with respect to ionic dissociation below 100 K at the surface of ASW. The slight decrease in ionic dissociation propensity reported for nitric acid at liquid water surfaces thus appears to be overwhelmed by the strong exothermicity of this reaction as its entropic inhibition becomes increasingly suppressed the lower the temperature. These findings may be relevant for atmospheric chemistry processes involving nitric acid, which should thus, according to thermodynamic considerations, be expected to behave as a strong acid at the surface of supercooled aerosols and of the quasi-liquid layer.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
2012
Marchand, Patrick; Marcotte, Guillaume; Ayotte, Patrick
Spectroscopic Study of HNO3 Dissociation on Ice Article de journal
Dans: J. Phys. Chem. A, vol. 116, no. 49, p. 12112–12122, 2012, ISSN: 1520-5215.
@article{Marchand2012,
title = {Spectroscopic Study of HNO_{3} Dissociation on Ice},
author = {Patrick Marchand and Guillaume Marcotte and Patrick Ayotte},
doi = {10.1021/jp309533f},
issn = {1520-5215},
year = {2012},
date = {2012-12-13},
urldate = {2012-12-13},
journal = {J. Phys. Chem. A},
volume = {116},
number = {49},
pages = {12112--12122},
publisher = {American Chemical Society (ACS)},
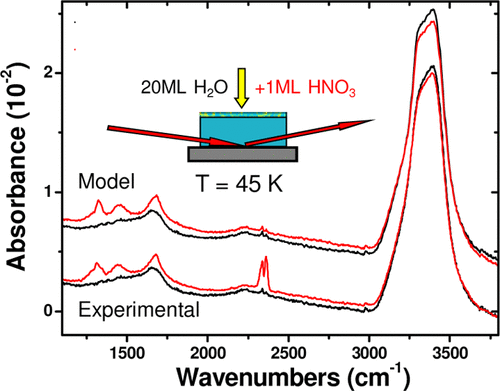
abstract = {A detailed spectroscopic study of HNO3:H2O binary amorphous mixtures, and of the adsorption of HNO3 onto ice, is reported. Using a classical optics model, the extent of intermixing and of ionic dissociation of adsorbed HNO3, which forms a strong acid with liquid water, is determined as a function of HNO3 coverage and temperature. Even at temperatures as low as 45 K, where intermixing is limited to at most a few molecular layers at the interface, ionic dissociation of adsorbed HNO3 is observed to be extensive. While some amount of molecularly adsorbed HNO3 is observed at the surface of ice at 45 K, its ionic dissociation occurs irreversibly upon heating the ice substrate to 120 K. The molecularly adsorbed state of HNO3 is not restored upon cooling, suggesting HNO3 is a metastable entity at the surface of ice. Therefore, despite ionic dissociation of HNO3 being thermodynamically favored, it appears to be kinetically inhibited at the surface of amorphous solid water at temperatures below 120 K.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Turgeon, Pierre-Alexandre; Ayotte, Patrick; Lisitsin, Elina; Meir, Yossi; Kravchuk, Tatyana; Alexandrowicz, Gil
Preparation, isolation, storage, and spectroscopic characterization of water vapor enriched in the ortho-H2O nuclear spin isomer Article de journal
Dans: Phys. Rev. A, vol. 86, no. 6, 2012, ISSN: 1094-1622.
@article{Turgeon2012,
title = {Preparation, isolation, storage, and spectroscopic characterization of water vapor enriched in the ortho-H_{2}O nuclear spin isomer},
author = {Pierre-Alexandre Turgeon and Patrick Ayotte and Elina Lisitsin and Yossi Meir and Tatyana Kravchuk and Gil Alexandrowicz},
doi = {10.1103/physreva.86.062710},
issn = {1094-1622},
year = {2012},
date = {2012-12-00},
urldate = {2012-12-00},
journal = {Phys. Rev. A},
volume = {86},
number = {6},
publisher = {American Physical Society (APS)},
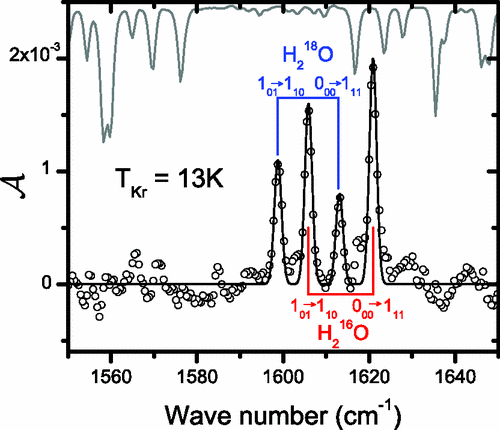
abstract = {Using magnetic focusing in a supersonic jet, a beam of “normal” H2O molecules seeded in a krypton carrier gas is shown to provide a source of water molecules that is highly enhanced in the ortho-H2O (𝑜-H2O) nuclear spin isomer over the high-temperature equilibrium 3:1 ortho:para ratio. Water from the magnetically focused beam is then isolated and stored within a Kr matrix at 13 K whereby the amplitude and lifetime of this strong nuclear spin polarization are quantified spectroscopically. Attempts to store the polarization in a colder Kr matrix reveal complex nuclear spin conversion processes.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
2011
Ayotte, Patrick; Marchand, Patrick; Daschbach, John L.; Smith, R. Scott; Kay, Bruce D.
HCl Adsorption and Ionization on Amorphous and Crystalline H2O Films below 50 K Article de journal
Dans: J. Phys. Chem. A, vol. 115, no. 23, p. 6002–6014, 2011, ISSN: 1520-5215.
@article{Ayotte2011,
title = {HCl Adsorption and Ionization on Amorphous and Crystalline H_{2}O Films below 50 K},
author = {Patrick Ayotte and Patrick Marchand and John L. Daschbach and R. Scott Smith and Bruce D. Kay},
doi = {10.1021/jp110398j},
issn = {1520-5215},
year = {2011},
date = {2011-06-16},
urldate = {2011-06-16},
journal = {J. Phys. Chem. A},
volume = {115},
number = {23},
pages = {6002--6014},
publisher = {American Chemical Society (ACS)},
abstract = {Molecular beams were used to grow amorphous and crystalline H2O films and to dose HCl upon their surface. The adsorption state of HCl on the ice films was probed with infrared spectroscopy. A Zundel continuum is clearly observed for exposures up to the saturation HCl coverage on ice upon which features centered near 2530, 2120, 1760, and 1220 cm−1 are superimposed. The band centered near 2530 cm−1 is observed only when the HCl adlayer is in direct contact with amorphous solid water or crystalline ice films at temperatures as low as 20 K. The spectral signature of solid HCl (amorphous or crystalline) was identified only after saturation of the adsorption sites in the first layer or when HCl was deposited onto a rare gas spacer layer between the HCl and ice film. These observations strongly support conclusions from recent electron spectroscopy work that reported ionic dissociation of the first layer HCl adsorbed onto the ice surface is spontaneous.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Marcotte, Guillaume; Ayotte, Patrick
Dans: vol. 134, no. 11, 2011, ISSN: 1089-7690.
@article{Marcotte2011,
title = {Strong intermolecular coupling between the HF stretching and H_{2}O bending vibrations in HF:H_{2}O binary amorphous solids: Breakdown of the electrostatic description of the hydrogen bond},
author = {Guillaume Marcotte and Patrick Ayotte},
doi = {10.1063/1.3562368},
issn = {1089-7690},
year = {2011},
date = {2011-03-21},
urldate = {2011-03-21},
volume = {134},
number = {11},
publisher = {AIP Publishing},
abstract = {The coupling mechanism between the HF stretching and H2O bending vibrations observed in the infrared spectra of HF:H2O binary amorphous solids is analyzed using a simple cluster model. The intermolecular vibrational coupling derived from electrostatic potentials is one order of magnitude smaller, and of the opposite sign, than that obtained from electronic structure-based potentials. This highlights the distinctively covalent character of strong H-bonds and unveils fundamental weaknesses of electrostatic descriptions of vibrational energy transfer in liquid water and aqueous solutions.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
2009
Ayotte, Patrick; Rafiei, Zohreh; Porzio, François; Marchand, Patrick
Dissociative adsorption of hydrogen fluoride onto amorphous solid water Article de journal
Dans: vol. 131, no. 12, 2009, ISSN: 1089-7690.
@article{Ayotte2009,
title = {Dissociative adsorption of hydrogen fluoride onto amorphous solid water},
author = {Patrick Ayotte and Zohreh Rafiei and François Porzio and Patrick Marchand},
doi = {10.1063/1.3231999},
issn = {1089-7690},
year = {2009},
date = {2009-09-28},
urldate = {2009-09-28},
volume = {131},
number = {12},
publisher = {AIP Publishing},
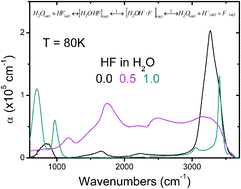
abstract = {Adsorption of hydrogen fluoride (HF) onto amorphous solid water films at 50 K is reported to yield a strong absorbance continuum in their reflection-absorption infrared spectra (RAIRS). This and other complex features observed in the RAIRS spectra of stratified binary composite HF:H2O nanoscopic films deposited onto Pt(111) are interpreted quantitatively using a classical optics model. Comparison with experimental data allows us to determine that the absorbance continuum is due to absorption within the film (as opposed to trivial optical effects) and that the extent of intermixing and uptake is mostly limited to the first few molecular layers. Furthermore, extensive isotope scrambling is demonstrated by the observation of similar Zundel continua upon codeposition of neat HF, or DF, and H2O vapors onto Pt(111) at 50 K. These observations are consistent with those expected from extensive ionic dissociation of HF upon dissolution within, and adsorption onto, ASW at 50 K.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Cholette, François; Zubkov, Tykhon; Smith, R. Scott; Dohnálek, Zdenek; Kay, Bruce D.; Ayotte, Patrick
Infrared Spectroscopy and Optical Constants of Porous Amorphous Solid Water Article de journal
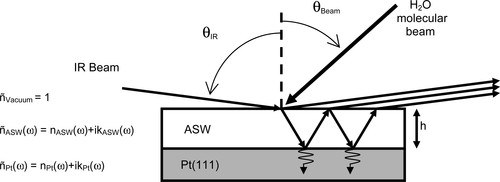
Dans: J. Phys. Chem. B, vol. 113, no. 13, p. 4131–4140, 2009, ISSN: 1520-5207.
@article{Cholette2008,
title = {Infrared Spectroscopy and Optical Constants of Porous Amorphous Solid Water},
author = {François Cholette and Tykhon Zubkov and R. Scott Smith and Zdenek Dohnálek and Bruce D. Kay and Patrick Ayotte},
doi = {10.1021/jp806738a},
issn = {1520-5207},
year = {2009},
date = {2009-04-02},
urldate = {2009-04-02},
journal = {J. Phys. Chem. B},
volume = {113},
number = {13},
pages = {4131--4140},
publisher = {American Chemical Society (ACS)},
abstract = {Reflection−absorption infrared spectra (RAIRS) of amorphous solid water (ASW) films grown at 20 K on a Pt(111) substrate at various angles (θBeam= 0−85°) using a molecular beam are reported. They display complex features arising from the interplay between refraction, absorption within the sample, and interference effects between the multiple reflections at the film−substrate and film−vacuum interfaces. Using a simple classical optics model based on Fresnel equations, we obtain optical constants [i.e., n(ω) and k(ω)] for porous ASW in the 1000−4000 cm−1 (10−2.5 μm) range. The behavior of the optical properties of ASW in the intramolecular OH stretching region with increasing θBeam is shown to be strongly correlated with its decreasing density and increasing surface area. A direct comparison between the RAIRS and calculated vibrational spectra shows a large difference (∼200 cm−1) in the position of the coupled H-bonded intramolecular OH stretching vibrations spectral feature. Moreover, this band shifts in opposite directions with increasing θBeam in RAIRS and vibrational spectra demonstrating RAIRS spectra cannot be interpreted straightforwardly as vibrational spectra due to severe optical distortions from refraction and interference effects.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
2008
Iftimie, Radu; Thomas, Vibin; Plessis, Sylvain; Marchand, Patrick; Ayotte, Patrick
Spectral Signatures and Molecular Origin of Acid Dissociation Intermediates Article de journal
Dans: J. Am. Chem. Soc., vol. 130, no. 18, p. 5901–5907, 2008, ISSN: 1520-5126.
@article{Iftimie2008,
title = {Spectral Signatures and Molecular Origin of Acid Dissociation Intermediates},
author = {Radu Iftimie and Vibin Thomas and Sylvain Plessis and Patrick Marchand and Patrick Ayotte},
doi = {10.1021/ja077846o},
issn = {1520-5126},
year = {2008},
date = {2008-05-01},
urldate = {2008-05-01},
journal = {J. Am. Chem. Soc.},
volume = {130},
number = {18},
pages = {5901--5907},
publisher = {American Chemical Society (ACS)},
abstract = {The existence of a broad, mid-infrared absorption ranging from 1000 to 3000 cm−1 is usually interpreted as a signature for the existence of protonated water networks. Herein, we use cryogenic mixtures of water and hydrogen fluoride (HF) and show experimental and computational evidence that similarly wide absorptions can be generated by a broad distribution of proton-shared and ion pair complexes. In the present case, we demonstrate that the broadening is mainly inhomogeneous, reflecting the fact that the topology of the first solvation shell determines the local degree of ionization and the shared-proton asymmetric stretching frequency within H2O·HF complexes. The extreme sensitivity of the proton transfer potential energy hypersurface to local hydrogen bonding topologies modulates its vibrational frequency from 2800 down to ∼1300 cm−1, the latter value being characteristic of solvation geometries that yield similar condensed-phase proton affinities for H2O and fluoride. By linking the local degree of ionization to the solvation pattern, we are able to propose a mechanism of ionization for HF in aqueous solutions and to explain some of their unusual properties at large concentrations. However, an important conclusion of broad scientific interest is our prediction that spectral signatures that are normally attributed to protonated water networks could also reveal the presence of strong hydrogen bonds between un-ionized acids and water molecules, with important consequences to spectroscopic investigations of biologically relevant proton channels and pumps.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Ayotte, Patrick; Plessis, Sylvain; Marchand, Patrick
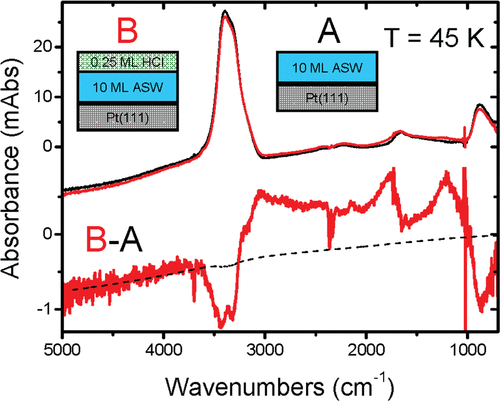
Trapping proton transfer intermediates in the disordered hydrogen-bonded network of cryogenic hydrofluoric acid solutions Article de journal
Dans: Phys. Chem. Chem. Phys., vol. 10, no. 32, 2008, ISSN: 1463-9084.
@article{Ayotte2008b,
title = {Trapping proton transfer intermediates in the disordered hydrogen-bonded network of cryogenic hydrofluoric acid solutions},
author = {Patrick Ayotte and Sylvain Plessis and Patrick Marchand},
doi = {10.1039/b806654j},
issn = {1463-9084},
year = {2008},
date = {2008-00-00},
urldate = {2008-00-00},
journal = {Phys. Chem. Chem. Phys.},
volume = {10},
number = {32},
publisher = {Royal Society of Chemistry (RSC)},
abstract = {A molecular-level description of the structural and dynamical aspects that are responsible for the weak acid behaviour of dilute hydrofluoric acid solutions and their unusual increased acidity at near equimolar concentrations continues to elude us. We address this problem by reporting reflection–absorption infrared spectra (RAIRS) of cryogenic HF–H2O binary mixtures at various compositions prepared as nanoscopic films using molecular beam techniques. Optical constants for these cryogenic solutions [n(ω) and k(ω)] are obtained by iteratively solving Fresnel equations for stratified media. Modeling of the experimental RAIRS spectra allow for a quantitative interpretation of the complex interplay between multiple reflections, optical interference and absorption effects. The evolution of the strong absorption features in the intermediate 1000–3000 cm−1 range with increasing HF concentration reveals the presence of various ionic dissociation intermediates that are trapped in the disordered H-bonded network of cryogenic hydrofluoric acid solutions. Our findings are discussed in light of the conventional interpretation of why hydrofluoric acid is a weak acid revealing molecular-level details of the mechanism for HF ionization that may be relevant to analogous elementary processes involved in the ionization of weak acids in aqueous solutions.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
2006
Marchand, Patrick; Riou, Samuel; Ayotte, Patrick
Diffusion Kinetics for Methanol in Polycrystalline Ice Article de journal
Dans: J. Phys. Chem. A, vol. 110, no. 41, p. 11654–11664, 2006, ISSN: 1520-5215.
@article{Marchand2006,
title = {Diffusion Kinetics for Methanol in Polycrystalline Ice},
author = {Patrick Marchand and Samuel Riou and Patrick Ayotte},
doi = {10.1021/jp0640878},
issn = {1520-5215},
year = {2006},
date = {2006-10-01},
urldate = {2006-10-01},
journal = {J. Phys. Chem. A},
volume = {110},
number = {41},
pages = {11654--11664},
publisher = {American Chemical Society (ACS)},
abstract = {Quantitative analyses of the isothermal desorption kinetics from methanol-doped H2O films on Pt(111) reveal that transport kinetics for CH3OH in polycrystalline ice are much slower than previously reported. They also indicate that MeOH displays first-order desorption kinetics with respect to its instantaneous surface concentration below 0.1 mole fraction in ice. These observations allow isothermal desorption rate measurements to be interpreted in terms of a depth profiling analysis providing one-dimensional concentration depth profiles from methanol-doped polycrystalline ice films. Using a straightforward approach to inhibit ice sublimation, transport properties are extracted from the evolution of concentration depth profiles obtained after thermal annealing of binary ice films at high temperature. Heterodiffusion coefficients for methanol in polycrystalline (cubic) ice Ic films are reported for temperatures between 145 and 195 K and for concentrations below 10-3 mole fraction. Finally, diffusion kinetics for methanol in ice are shown to display a very strong concentration dependence that may contribute, in addition to variations in laboratory samples microstructure, to the disagreements reported in the literature regarding the transport properties of ice.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
2005
Ayotte, Patrick; Hébert, Martin; Marchand, Patrick
Why is hydrofluoric acid a weak acid? Article de journal
Dans: vol. 123, no. 18, 2005, ISSN: 1089-7690.
@article{Ayotte2005,
title = {Why is hydrofluoric acid a weak acid?},
author = {Patrick Ayotte and Martin Hébert and Patrick Marchand},
doi = {10.1063/1.2090259},
issn = {1089-7690},
year = {2005},
date = {2005-11-08},
urldate = {2005-11-08},
volume = {123},
number = {18},
publisher = {AIP Publishing},
abstract = {The infrared vibrational spectra of amorphous solid water thin films doped with HF at 40K reveal a strong continuous absorbance in the 1000–3275cm−1 range. This so-called Zundel continuum is the spectroscopic hallmark for aqueous protons. The extensive ionic dissociation of HF at such low temperature suggests that the reaction enthalpy remains negative down to 40K. These observations support the interpretation that dilute HF aqueous solutions behave as weak acids largely due to the large positive reaction entropy resulting from the structure making character of the hydrated fluoride ion.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}